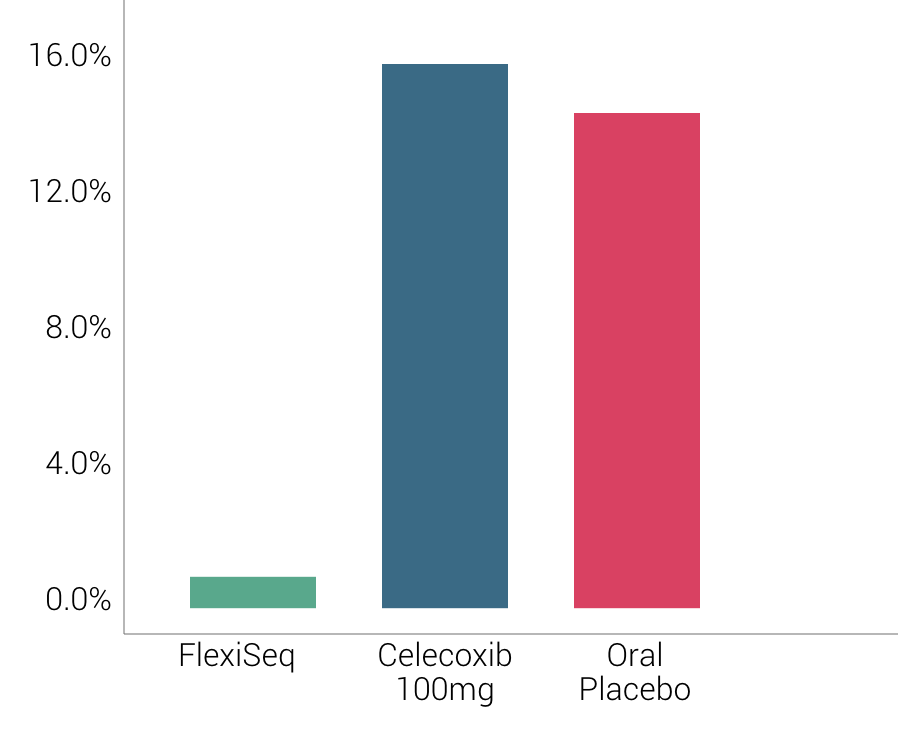About FlexiSEQ
Research on joint disorders
Patients with joint disorders such as osteoarthritis have been found to have reduced levels of synovial lubricants that play a key role in protecting articulating cartilage. Research has shown that liposomal aggregates that are highly compressible, soft and are highly hydrated make effective biolubricants. A study with topically applied, fluorescently labelled Sequessome vesicles showed that the Sequessomes travel to the synovium, where they arrive intact and distribute themselves on the surface of the cartilage, forming a protective, lubricating layer.
FlexiSEQ efficacy
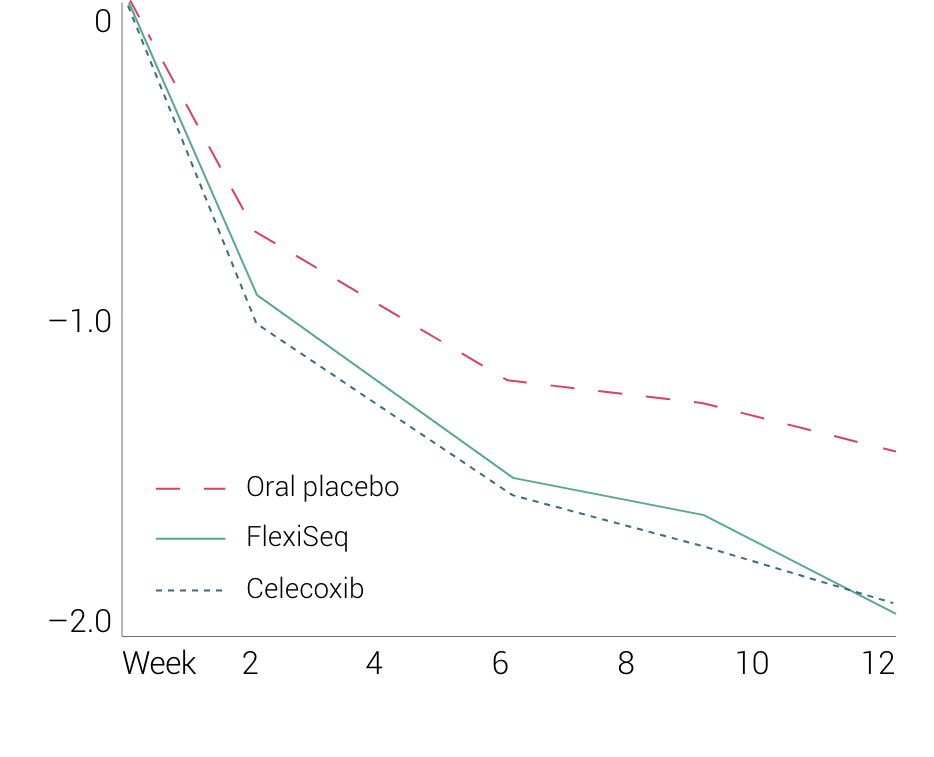
By exploiting this lubricating mechanism, FlexiSEQ demonstrated clear efficacy in a large scale, Phase III, 12-week trial, compared to the oral selective, COX2 painkiller, celecoxib. These data are shown below:
FlexiSEQ safety profile
The excellent safety profile has been further demonstrated by the total lack of any systemic side effects being related to the product in over 90 million doses sold worldwide.
What healthcare professionals are saying about FlexiSEQ
The advantages of FlexiSEQ have been recognised by key persons within the arthritis medical community and leading physio-therapy/sports rehabilitation:
“There are fewer and fewer safe, available, pain-relieving options for people with osteoarthritis. If this product can relieve their long-term pain without side-effects, then that’s good news for the millions of people who suffer with this debilitating condition.” Arthritis Research UK.
“This is a fantastic product and one the medical team are proud and happy to have associated with the Club” – Joe Collins, former Head of Saracens Medical Team and now High Performance Manager of Buffalo Sabres (NHL) and Director of Performance Science of Buffalo Bills (NFL).
FlexiSEQ trial data have been published in a number of journals: Current Medical Research and Opinion, Rheumatology, Journal of Rheumatology, and Journal of Pain Research.